Describe bohr's model of the atom3/15/2024 According to Heisenberg’s principle simultaneous measurement of position ans momentum of a small moving particle is impossible.Therefore, well-defined stationary orbits cannot exist.Bohr treated electron as a particle which is contrary to de Broglie’s dual nature of electron.Now, an atom that is in the state of the smallest orbit will be. The reason for atoms being unstable is that electrons jump to lower orbits causing radiation. Therefore, smaller orbits will have lower energy electrons. The word ‘atom’ actually comes from Ancient Greek and roughly translates as. To describe Bohrs atomic model, it can be said that the energy levels of the electrons concentrate on the size of the orbits. In fact, we have to go all the way back to Ancient Greece to find its genesis. Though our graphic starts in the 1800s, the idea of atoms was around long before. It couldnot explain the splitting of spectral lines in the presence of magnetic field. This graphic takes a look at the key models proposed for the atom, and how they changed over time.Bohrs model suggests that each atom has a set of unchangeable energy levels, and electrons in the electron cloud of that atom must be in one of those energy levels. It couldnot explain the highly resolved spectra of hydrogen and hydrogen like atoms which show a fine structure. 2.12: The Bohr Model - Atoms with Orbits is shared under a CK-12 license and was authored, remixed, and/or curated by Marisa Alviar-Agnew & Henry Agnew.It cannot explain the spectra of multielectron elements.He gave a satisfactory explaination for the spectra of hydrogen and hydrogen like species. He told that electrons revolve around the nucleus in specified circular paths called as orbits and each orbit is associated with a definite amount of energy. According to this theory, the electrons should lose energy continuously and hence electron will eventually fall into the nucleus. He proposed that electrons revolve around the nucleus like planets revolve around the sun. S2CID 250900220.Rutherford was unable to explain the stability of atom. "Sommerfeld formula and Dirac's theory" (PDF). ^ - Atombau und Spektrallinien, 1921, page 520 How did scientists figure out the structure of atoms without looking at them Try out different models by shooting light at the atom.All features of Bohr’s model of the atom can be summarized in Bohr’s Postulates. "The Kossel-Sommerfeld Theory and the Ring Atom". In atomic physics, the Bohr model of the atom (also known as the Rutherford-Bohr model) is the modern model of the hydrogen atom introduced by Danish physicist Niels Bohr. "Einstein's unknown insight and the problem of quantizing chaos" (PDF). Learn the basic concept of Bohrs model of the atom, which states that electrons revolve around the nucleus in fixed orbits with different energy levels or quantum numbers. ^ The Collected Papers of Albert Einstein, vol. 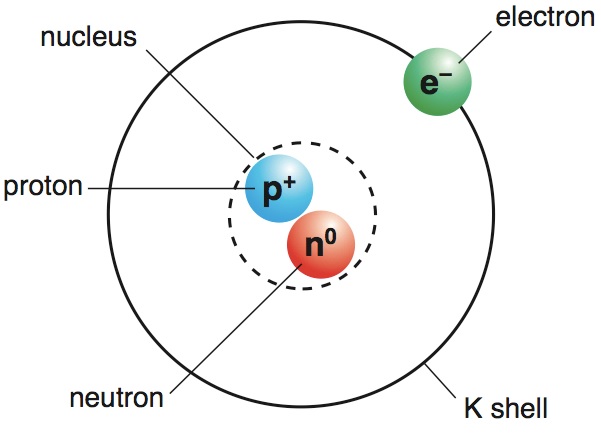
(iii) All the protons and neutrons of the atom are contained in the nucleus. (ii) The whole mass of the atom is concentrated in the nucleus and the volume of the nucleus is smaller than the volume of the atom by a ratio of about 1 : 10 5. Therefore, these circular orbits are also known as energy levels or energy shells. Each shell or orbit corresponds to a definite energy. In an atom, the electrons revolve around the nucleus in certain definite circular paths called orbits, or shells. "The quantum theory of radiation and line spectra". According to Bohrs theory: (i) The atom consists of a small (positively charged) nucleus at its centre. Main postulates of Bohrs model of an atom are: 1. 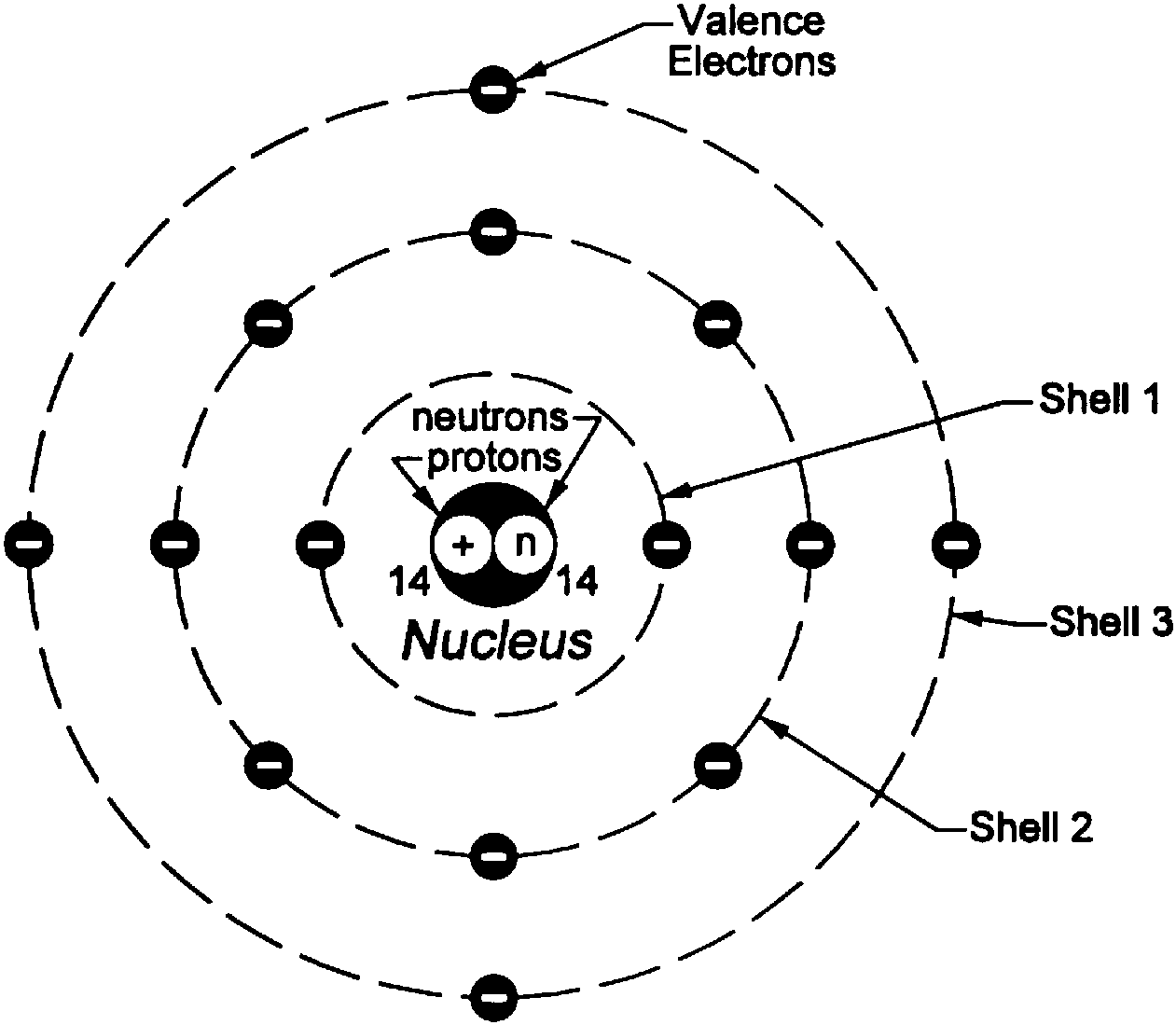
Nevertheless, both solutions fail to predict the Lamb shifts. This solution (using substitutions for quantum numbers) is equivalent to the solution of the Dirac equation. What was thought of as a single particle about 1 ×. The idea of the atom as the building block of matter has developed over time. The Bohr–Sommerfeld model supplemented the quantized angular momentum condition of the Bohr model with an additional radial quantization condition, the Wilson– Sommerfeld quantization condition ∫ 0 T p r d q r = n h, is the fine-structure constant. GCSE AQA Trilogy Models of the atom - AQA Developing the atom. Sommerfeld argued that if electronic orbits could be elliptical instead of circular, the energy of the electron would be the same, except in the presence of a magnetic field, introducing what is now known as quantum degeneracy. Bohr–Sommerfeld theory is named after Danish physicist Niels Bohr and German physicist Arnold Sommerfeld. The Bohr–Sommerfeld model (also known as the Sommerfeld model or Bohr–Sommerfeld theory) was an extension of the Bohr model to allow elliptical orbits of electrons around an atomic nucleus. The Sommerfeld extensions of the 1913 solar system Bohr model of the hydrogen atom showing the addition of elliptical orbits to explain spectral fine structure.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
